AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Subchondral sclerosis8/14/2023  
Because edema has not been shown to contribute substantially to subchondral pathology and is not necessary to diagnose pathologic bone marrow changes either radiographically or developmentally, ‘bone marrow edema’ is a misleading term. īML were originally known as ‘bone marrow edema’, a term originally coined in 1988 to describe the characteristic MRI changes. But while BML do correlate with hip pain and joint space narrowing, this pathology has been far less studied than its counterpart at the knee. In the knee, BML have been reliably shown to predate these sequelae of OA. 
BML remain a key feature of OA and have strong associations with pain, cartilage defects, and indicated joint replacement. Throughout life, yellow (inactive) bone marrow predictably replaces red (active) bone marrow however, the appearance of BML on MRI is a departure from this easily identifiable and non-pathologic process of marrow replacement. This review will summarize the etiology, pathogenesis, radiographic findings, classification and treatment associated with BML and OA of the hip. 
BML in the knee have been increasingly studied however, due to the hip’s unique biomechanical architecture, function and loading, independent risk factors leading to hip OA, as well as the association between BML and multiple non-osteoarthritic diseases, hip BMLs must be independently understood. Due to their predictable classification patterns on MRI, BML may become targets of future therapies to treat or delay progression of hip OA. While they tend to occur in patients with reduced hip cartilage volume, BML also may be found as a precursor to cartilage depletion or less commonly in asymptomatic patients without OA of the hip. The BML, one such subchondral pathology, has been routinely identified in patients with symptomatic hip OA. It was then suggested that OA holds some association with underlying joint pathologies, including abnormal loading forces, abnormal articular cartilage or defective subchondral support. Using detailed evaluation of femoral head radiographs, Solomon classified osteoarthritic hips based on the presence of characteristic anatomical features, destructive changes and repair phenomena. Bone marrow lesions (BML), areas of altered magnetic resonance imaging signal within subchondral bone, are commonly identified in osteoarthritic patients however, BML remain poorly understood, especially about the hip. While cartilage damage in OA has been well studied, the contributions of subchondral bone to this disease process have been explored only recently. One of the most prevalent musculoskeletal diseases and causes of lower extremity pain, osteoarthritis (OA) is characterized by discomfort, disability and morbidity. By continuing to define and refine the relationships between BML, subchondral bone cysts and OA, prevention, diagnosis and treatment of OA could shift, leading to an improved quality of life and increased longevity of individuals’ native hips. The identification of BML in the setting of a pre-osteoarthritic condition may provide a target for treatment and prevention of joint degeneration. Due to the hip’s unique biomechanical architecture, function and loading, and independent risk factors leading to hip OA, hip BMLs must be independently understood. While BML are known to be a common pathology throughout the body, BML at the hip have not been extensively studied in comparison to those at the knee. Created via repetitive microdamage at the articular surface and dysregulated subchondral healing, BML have been linked to traumatic, inflammatory, degenerative, metabolic and neoplastic processes. doi:10.1002/cdt3.16.Bone marrow lesions (BML) are painful changes in subchondral bone which can be reliably identified on magnetic resonance imaging and have been identified in patients suffering from hip osteoarthritis (OA) and related conditions. Sex, but not age and bone mass index positively impact on the development of osteochondral micro-defects and the accompanying cellular alterations during osteoarthritis progression. Kaspiris A, Chronopoulos E, Vasiliadis E, Khaldi L, Melissaridou D, Iliopoulos ID, et al. Subchondral bone remodelling in osteoarthritis. Subchondral bone marrow lesions are highly associated with, and predict subchondral bone attrition longitudinally: the MOST study. 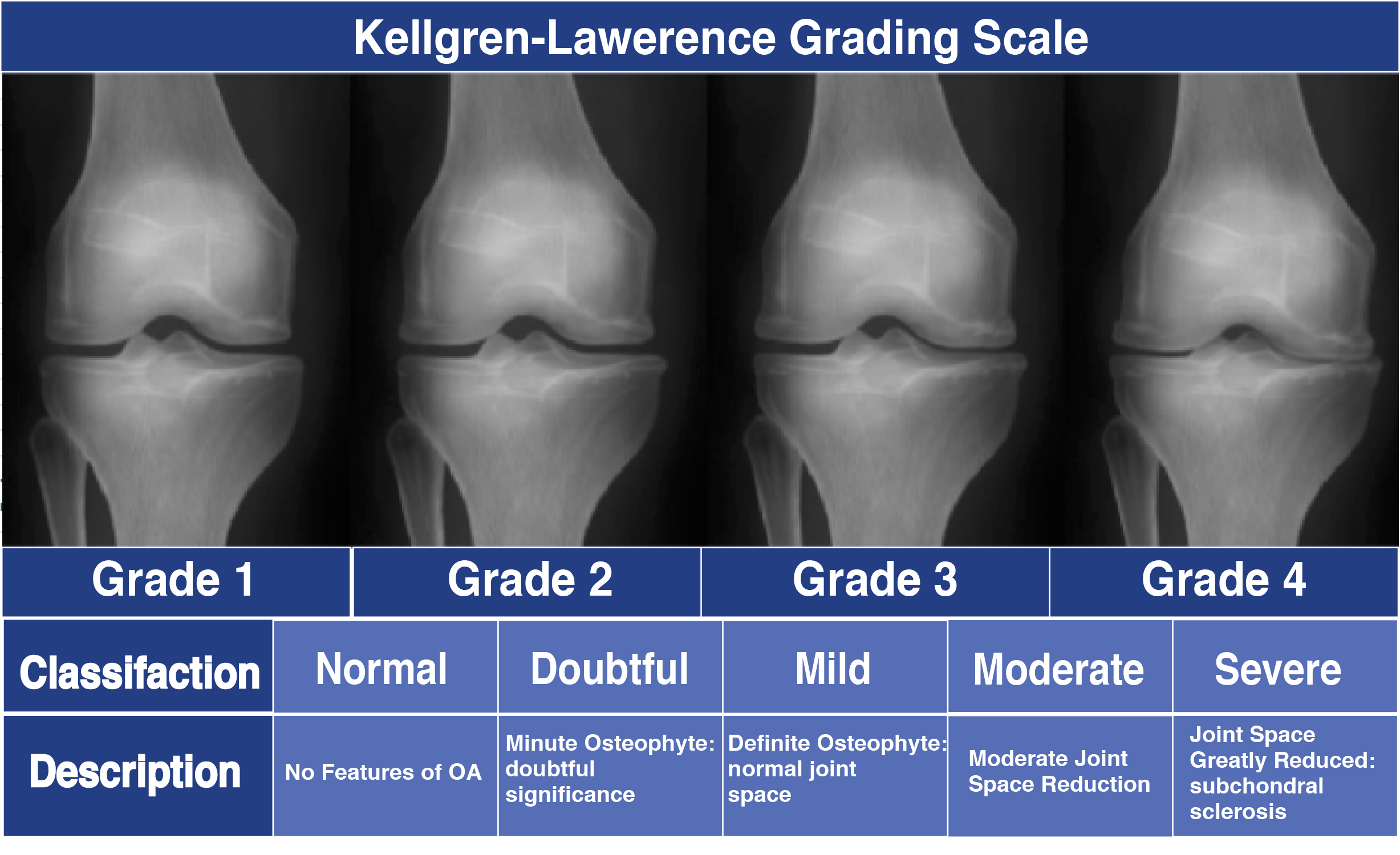
Insight into the potential pathogenesis of human osteoarthritis via single-cell RNA sequencing data on osteoblasts. Alpha C-telopeptide of type I collagen is associated with subchondral bone turnover and predicts progression of joint space narrowing and osteophytes in osteoarthritis. Huebner JL, Bay-Jensen AC, Huffman KM, et al. Subchondral bone in osteoarthritis: insight into risk factors and microstructural changes. Bone-cartilage crosstalk: a conversation for understanding osteoarthritis. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
